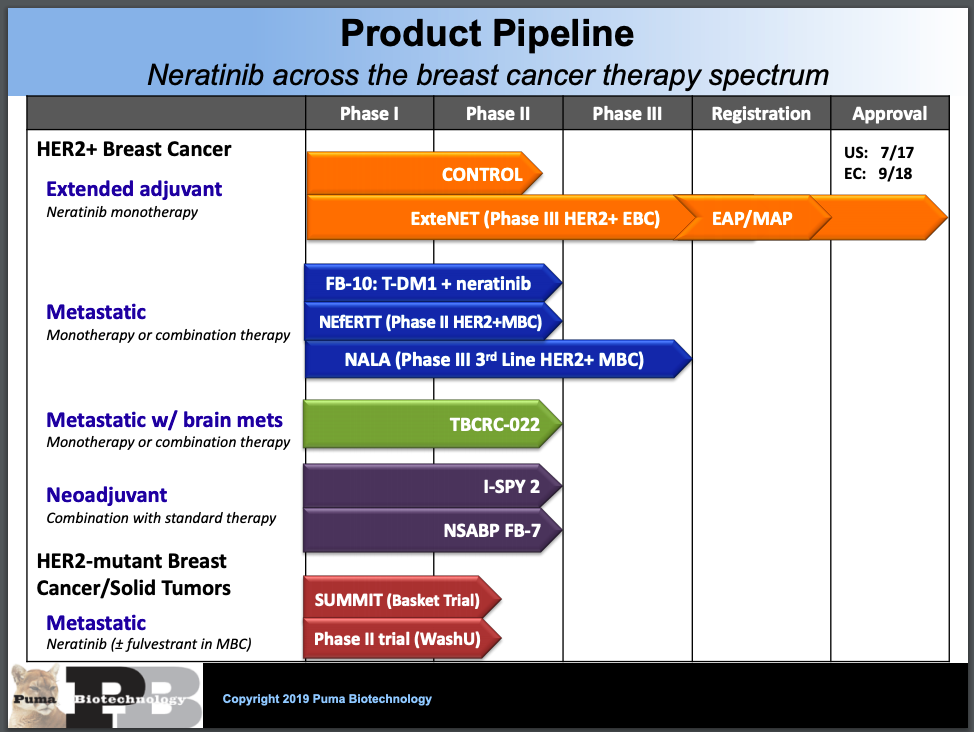
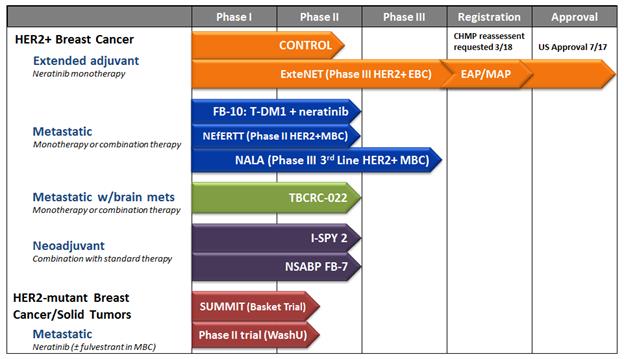
PUMA BIOTECHNOLOGY: Announces U.S. FDA Acceptance of Supplemental New Drug Application for Neratinib to Treat HER2-Positive Metastatic Breast Cancer | FDA Health News

Vir Biotechnology to Provide Corporate Update and Report Third Quarter 2022 Financial Results on November 3, 2022
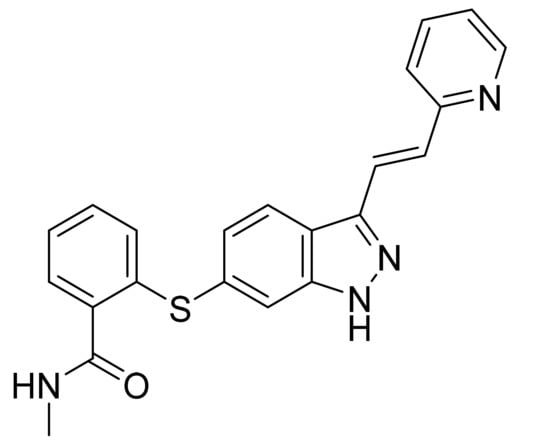
Pharmaceuticals | Free Full-Text | Innovations and Patent Trends in the Development of USFDA Approved Protein Kinase Inhibitors in the Last Two Decades | HTML