The Interactions of the 5-HT3 Receptor with Quipazine-Like Arylpiperazine Ligands. The Journey Track at the End of the First Dec
Design, Synthesis, and Biological Evaluation of AT1 Angiotensin II Receptor Antagonists Based on the Pyrazolo[3,4-b]pyridine and

PDF) Synthesis and characterization of charge-transporting π-stacked polybenzofulvene derivatives | Marco Paolino - Academia.edu

PDF) Bithiophene-Based Polybenzofulvene Derivatives with High Stacking and Hole Mobility | Marco Paolino - Academia.edu

PDF) New π-stacked benzofulvene polymer showing thermoreversible polymerization: Studies in macromolecular and aggregate structures and polymerization mechanism | Maurizio Anzini - Academia.edu
Design, synthesis, structure, and photophysical features of highly emissive cinnamic derivatives - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D0NJ02429E
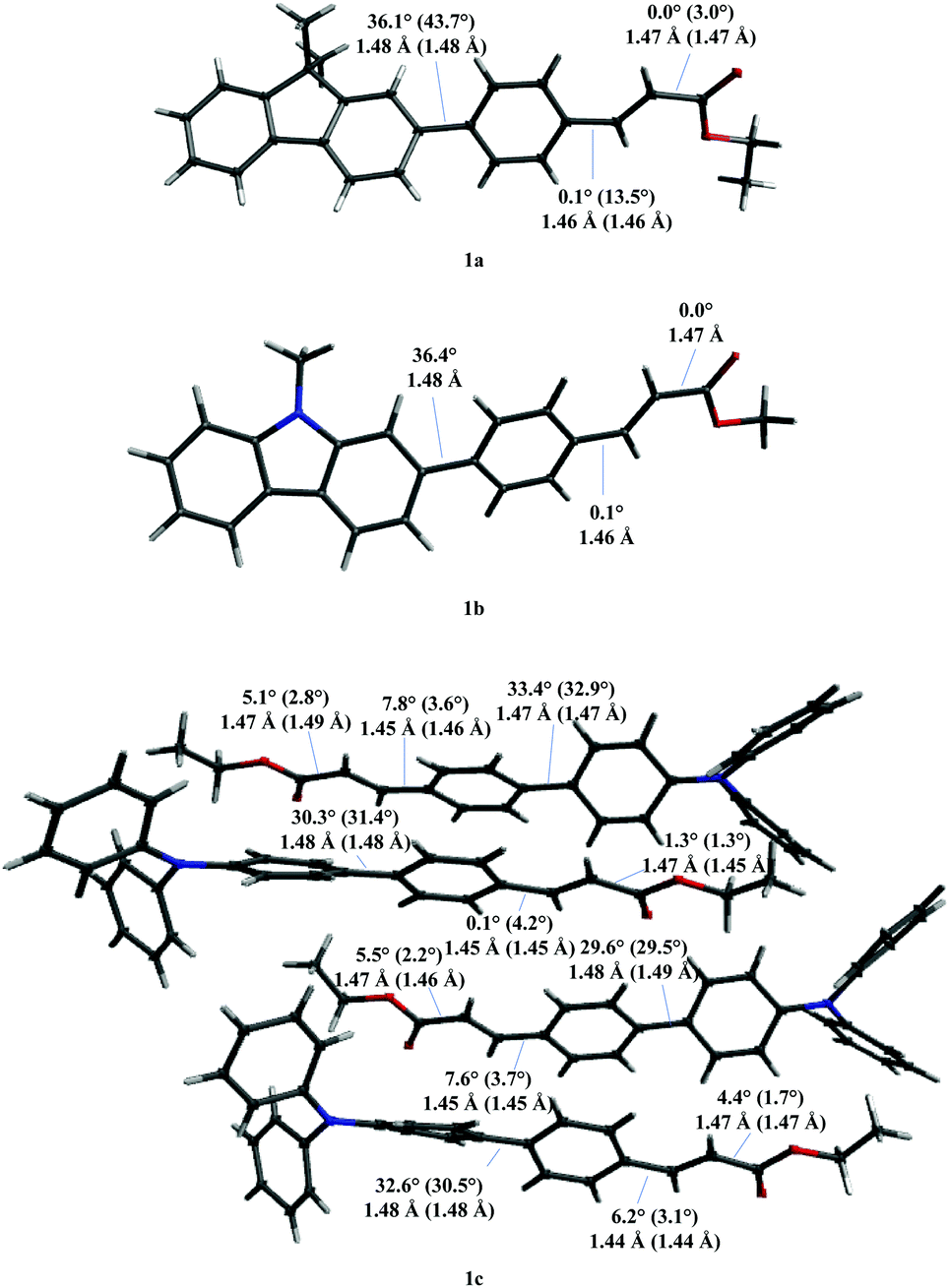
Design, synthesis, structure, and photophysical features of highly emissive cinnamic derivatives - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D0NJ02429E

Exploring Translocator Protein (TSPO) Medicinal Chemistry: An Approach for Targeting Radionuclides and Boron Atoms to Mitochondria | Journal of Medicinal Chemistry
Hyaluronan Graft Copolymers Bearing Fatty-Acid Residues as Self-Assembling Nanoparticles for Olanzapine Delivery
Structural Manipulation of the Conjugated Phenyl Moiety in 3-Phenylbenzofulvene Monomers: Effects on Spontaneous Polymerization

Xanthopsin‐Like Systems via Site‐Specific Click‐Functionalization of a Retinoic Acid Binding Protein - Tassone - 2022 - ChemBioChem - Wiley Online Library

PDF) Design, Synthesis and Characterization of a New Visible Light‐Sensitive Molecular Switch and its PEGylation Towards a Self‐Assembling Molecule

Federico GALVAGNI | Professor (Associate) | PhD | Università degli Studi di Siena, Siena | UNISI | Department of Biotechnology, Chemistry and Pharmaceutical Sciences

Design, synthesis, structure, and photophysical features of highly emissive cinnamic derivatives - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D0NJ02429E

Andrea CAPPELLI | Professor (Associate) | Università degli Studi di Siena, Siena | UNISI | Department of Biotechnology, Chemistry and Pharmaceutical Sciences | Professional profile







